Pharmacovigilance Market Forecast at US$ 15.7 Bn by 2033: Persistence Market Research
Global pharmacovigilance market driven by drug safety demands, regulatory compliance, outsourcing trends, and rising clinical research activities worldwide.
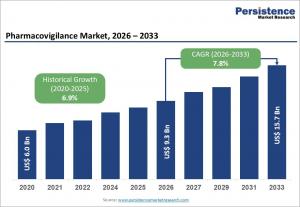
LONDON, UNITED KINGDOM, March 18, 2026 /EINPresswire.com/ -- The global pharmacovigilance market is witnessing significant expansion, driven by increasing emphasis on drug safety and regulatory compliance. The market size is estimated to reach US$ 9.3 billion in 2026 and is projected to grow to US$ 15.7 billion by 2033, registering a CAGR of 7.8% during the forecast period. This growth is primarily fueled by rising adverse drug reaction (ADR) reporting, growing drug approvals, and the need for continuous monitoring of drug safety throughout the product lifecycle. As pharmaceutical innovation accelerates, pharmacovigilance systems have become essential for ensuring patient safety and maintaining regulatory standards across global markets.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/36214
Key Industry Highlights
Spontaneous reporting remains the dominant segment, accounting for 41.2% of the market in 2025. Its importance lies in its ability to capture real-world safety data after drug commercialization. Regulatory authorities worldwide mandate reporting from healthcare professionals and patients, ensuring a steady flow of safety data for signal detection. This method forms the backbone of global pharmacovigilance systems due to its wide coverage and regulatory acceptance.
Regionally, North America leads the market with a 43.6% share in 2025, supported by strong regulatory frameworks, advanced healthcare infrastructure, and high drug approval rates. Meanwhile, Asia-Pacific is emerging as the fastest-growing region due to expanding pharmaceutical manufacturing, increasing clinical trials, and rising outsourcing activities in countries such as India and China.
Market Dynamics
Driver: Increasing Drug Approvals and Biologics Expansion
The growing number of drug approvals globally is a major driver of the pharmacovigilance market. As more therapies enter the market, especially complex biologics, the need for robust safety monitoring increases. Biologics, including monoclonal antibodies and advanced therapies, present unique safety challenges that require continuous surveillance. Regulatory agencies emphasize lifecycle safety monitoring, leading pharmaceutical companies to invest in advanced pharmacovigilance infrastructure. This surge in approvals results in higher volumes of adverse event reports and necessitates efficient data management systems, signal detection tools, and skilled professionals.
Restraint: High Operational Costs
Despite its importance, pharmacovigilance compliance involves substantial costs. Companies must invest in safety databases, software tools, skilled personnel, and regulatory reporting systems. Additionally, varying global regulatory requirements create complexity, requiring organizations to maintain multiple reporting standards. Small and mid-sized pharmaceutical companies often face challenges in managing these costs, which can limit their ability to scale pharmacovigilance operations effectively.
Opportunity: Outsourcing in Emerging Markets
Emerging markets present significant growth opportunities for pharmacovigilance outsourcing. Countries such as India, China, and Brazil offer cost-effective services, skilled workforce, and improving regulatory frameworks. Pharmaceutical companies increasingly rely on contract research organizations (CROs) in these regions to handle case processing, signal detection, and regulatory reporting. The adoption of advanced technologies such as cloud-based platforms and artificial intelligence further enhances the efficiency of outsourced services, making this a key growth area for the market.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/36214
Category-wise Analysis
By Reporting Type
Spontaneous reporting dominates due to its critical role in identifying ADRs in real-world settings. It provides extensive safety data and supports regulatory decision-making. The large volume of reports collected globally ensures continuous monitoring and early detection of potential safety issues.
By Deployment Mode
Outsourcing has become the preferred deployment model in pharmacovigilance. Pharmaceutical companies partner with specialized service providers to manage complex safety operations efficiently. Outsourcing allows access to global expertise, reduces operational burden, and ensures compliance with regulatory standards without significant internal investment.
Regional Insights
North America continues to dominate the pharmacovigilance market due to advanced regulatory systems, high adoption of digital health technologies, and strong pharmaceutical research activity. The region benefits from extensive ADR reporting systems and integration of electronic health records, enabling efficient safety monitoring.
Europe also plays a crucial role with its unified regulatory framework and standardized reporting systems. Strong collaboration among regulatory authorities ensures consistent safety monitoring across the region, contributing significantly to global pharmacovigilance practices.
Asia-Pacific is experiencing rapid growth due to expanding pharmaceutical industries, increasing clinical trials, and regulatory advancements. The region’s cost advantages and growing expertise in pharmacovigilance services are attracting global outsourcing activities, further accelerating market growth.
Competitive Landscape
The pharmacovigilance market is highly competitive, with key players focusing on technological innovation, strategic partnerships, and service expansion. Companies are investing in artificial intelligence and data analytics to enhance signal detection and automate case processing. Recent developments highlight the growing importance of AI-driven solutions and the increasing role of qualified pharmacovigilance professionals in managing complex safety systems.
Overall, the pharmacovigilance market is set for sustained growth, supported by regulatory demands, technological advancements, and the continuous expansion of the global pharmaceutical industry.
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/36214
Market Segmentation
By Product Type
Spontaneous Reporting
Intensified ADR Reporting
Targeted Spontaneous Reporting
Cohort Event Monitoring
EHR Mining
By Deployment Mode
In-house
Contract Outsourcing
By Product Life Cycle
Pre-clinical
Phase I
Phase II
Phase III
Phase IV
By Therapeutic Area
Oncology
Neurology
Infectious Diseases
Cardiology
Respiratory Systems
Others
By End-user
Pharmaceutical Companies
Contract Research Organizations (CROs)
Biotechnology Firms
Medical Device & Combination Product Manufacturers
Others
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East and Africa
Read Related Reports:
Custom-Compounded Therapy Market: Custom compounded therapy market to grow from US$6.1 Bn in 2026 to US$10.0 Bn by 2033 driven by chronic disease care and personalized medicine demand.
Oral Hydrogel Wound Dressing Market: Oral Hydrogel Wound Dressing Market to Reach US$1,459.4 Mn by 2033, Expanding at 8.4% CAGR from 2026 to 2033 Worldwide, Says Market Report.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.